Repurposing Ingenol Mebutate as a novel pharmaceutical therapy for normal and pathological dermal fibrosis
Introduction: Excessive dermal scarring/fibrosis poses major challenges to Healthcare Services worldwide. This is confounded by existing therapies being unsatisfactory at preventing or arresting fibrosis. Therefore, there is the significant need for novel anti-fibrotic therapies with improved efficacy. The anti-cancer drug, ingenol mebutate (IngM), is licensed for actinic keratosis and non-melanoma skin cancers. IngM also stimulates exceptional healing and reduced scarring in treated skin, implying IngM possesses potent anti-fibrotic properties. As transforming growth factor-β1 (TGF-β1)-driven, dermal fibroblast-myofibroblast differentiation is pivotal to scarring outcomes, fibroblasts and myofibroblasts represent viable IngM targets. This work evaluated IngM efficacy against normal (dermal) and pathological (keloid) fibroblasts/myofibroblasts; and its mechanisms of action.
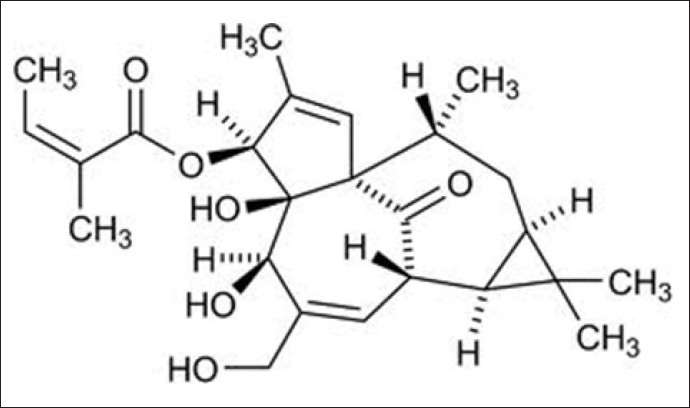
Materials and Methods: Human dermal and keloid fibroblasts were treated with IngM (0-10µg/mL) and rhTGF-β1 (10ng/mL); and assessed for myofibroblast differentiation by immunocytochemistry/QRT-PCR for a-smooth muscle actin (α-SMA) expression/stress fibre formation; QRT-PCR/Western blotting of other myofibroblast-/extracellular matrix (ECM)-related genes/proteins; hyaluronan synthesis/assembly (ELISA, hyaluronan synthase QRT-PCR, pericellular localization); and Microarrays/protein validation analysis.
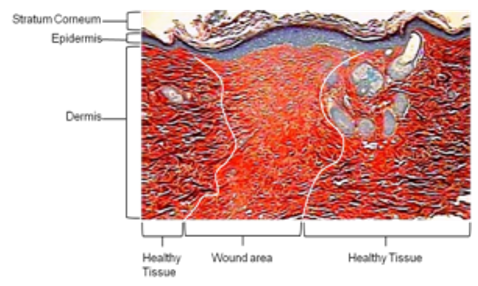
Findings: Although IngM does not inhibit dermal fibroblast-myofibroblast differentiation, IngM alters myofibroblast pro-fibrotic ECM deposition due to enhanced degradation of type I collagen by up-regulated matrix metalloproteinases (MMP-1, MMP-9). IngM also increases myofibroblast hyaluronan synthesis by hyaluronan synthase-2. Although reduced MMPs and hyaluronan are hallmarks of keloid scars, increased MMP/hyaluronan levels were also identified in IngM-treated keloid fibroblasts; in addition to reversed keloid fibroblast-myofibroblast formation and reduced type I collagen expression.
Conclusion: Therefore, IngM modulates dermal/keloid fibroblast differentiation and behavior, inducing scar tissue resolution. Such findings support IngM development/repurposing as a novel anti-fibrotic therapeutic against normal and pathological dermal scarring.
Author: Ryan Moseley, Cardiff University,
Conference: International Conference on Wound Care, Tissue Repair and Regenerative MedicineDate & venue: June 14-15, 2018 | London, UKWebsite: https://goo.gl/eLcFyZ

Materials and Methods: Human dermal and keloid fibroblasts were treated with IngM (0-10µg/mL) and rhTGF-β1 (10ng/mL); and assessed for myofibroblast differentiation by immunocytochemistry/QRT-PCR for a-smooth muscle actin (α-SMA) expression/stress fibre formation; QRT-PCR/Western blotting of other myofibroblast-/extracellular matrix (ECM)-related genes/proteins; hyaluronan synthesis/assembly (ELISA, hyaluronan synthase QRT-PCR, pericellular localization); and Microarrays/protein validation analysis.

Findings: Although IngM does not inhibit dermal fibroblast-myofibroblast differentiation, IngM alters myofibroblast pro-fibrotic ECM deposition due to enhanced degradation of type I collagen by up-regulated matrix metalloproteinases (MMP-1, MMP-9). IngM also increases myofibroblast hyaluronan synthesis by hyaluronan synthase-2. Although reduced MMPs and hyaluronan are hallmarks of keloid scars, increased MMP/hyaluronan levels were also identified in IngM-treated keloid fibroblasts; in addition to reversed keloid fibroblast-myofibroblast formation and reduced type I collagen expression.
Conclusion: Therefore, IngM modulates dermal/keloid fibroblast differentiation and behavior, inducing scar tissue resolution. Such findings support IngM development/repurposing as a novel anti-fibrotic therapeutic against normal and pathological dermal scarring.
Author: Ryan Moseley, Cardiff University,
Conference: International Conference on Wound Care, Tissue Repair and Regenerative MedicineDate & venue: June 14-15, 2018 | London, UKWebsite: https://goo.gl/eLcFyZ
Comments
Post a Comment